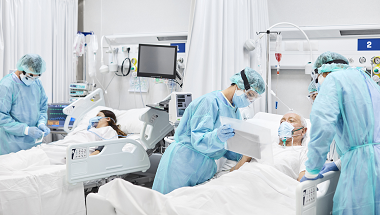
NICE updates managing COVID guideline with new monoclonal antibody recommendations
NICE has updated its managing COVID-19 guideline to include new recommendations on using monoclonal antibodies to treat COVID-19 patients in hospital.
All NICE products on COVID-19. Includes any guidance and advice.
We use the best available evidence to develop recommendations that guide decisions in health, public health and social care.
Critical assessment of evidence to help you make decisions. Advice, rather than formal NICE guidance.
Published advice on this topic (7)
Review the evidence across broad health and social care topics.
Reviews the clinical and cost-effectiveness of new treatments.
Review new diagnostic technologies for adoption in the NHS.
Reviews new medical devices for adoption in the NHS.
Review the evidence and likely costs of medical devices and technologies.
Guidance, quality standards and advice being developed.
Guidance and quality standards awaiting development.
Guidance in topic selection.
We also have resources to help local partnerships tackle their priorities. For advice specific to your area, contact your local NICE field team.
We've brought together guidance, resources and other information that may help you.
We've updated our summaries of current evidence for primary care professionals with some information about COVID-19. This is largely based on guidance from Public Health England.
We're part of the research to access pathway for investigational drugs for COVID-19 (RAPID-C19), a collaboration that aims to get treatments to patients quickly and safely.
These guides are aimed at specialists working in hospitals in England during the pandemic. They aim to help departments continue essential care for patients while supporting the wider hospital and community to provide care for people with COVID-19.
A guide for internal developers and external collaborators on assessing the quality of wider sources of data and evidence in our guidance.
A 3-stage approach to help anyone working on testing for COVID-19 to collect the best possible data and evidence while tests are being quickly developed and validated.
A guide to evidence collection for medicines, plus free scientific advice for researchers developing novel diagnostics or therapeutics for COVID-19.

NICE has updated its managing COVID-19 guideline to include new recommendations on using monoclonal antibodies to treat COVID-19 patients in hospital.

NICE has updated its advice on two therapeutics for managing COVID-19 – remdesivir and colchicine.
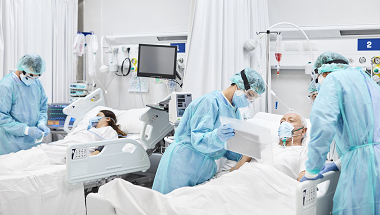
One year on from the first national lockdown, NICE has today (23 March) published a single guideline for the management of COVID-19 in children and adults.
Examples of how our guidance and standards have been put into practice in the NHS, local authorities, voluntary sector and a range of other organisations.
Showing 3 of 5 shared learning examples for this topic: