Overview
This guideline covers the diagnosis and management of tension-type headache, migraine (including migraine with aura and menstrual-related migraine), cluster headache and medication overuse headache in young people (aged 12 years and older) and adults. It aims to improve the recognition and management of headaches, with more targeted treatment to improve the quality of life for people with headaches, and to reduce unnecessary investigations.
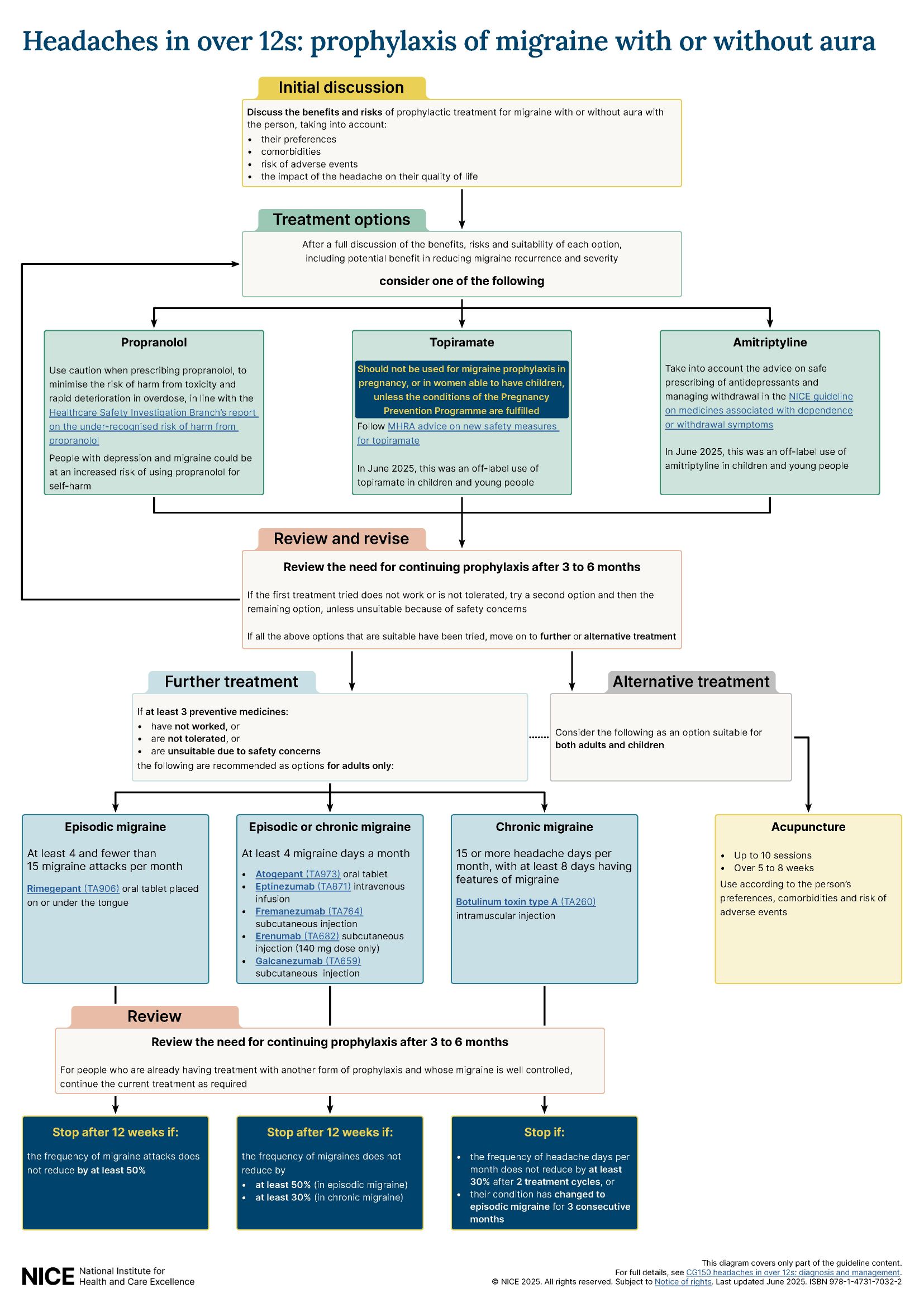
MHRA advice on topiramate: Recommendations on topiramate have taken into account the MHRA advice on the use of topiramate. Topiramate should not be used for migraine prophylaxis in pregnancy, or in women of childbearing potential unless the conditions of the Pregnancy Prevention Programme are fulfilled.
Last reviewed: 3 June 2025
We changed the strength of recommendations on migraine prevention to make the use of topiramate or propranolol a ‘consider’ recommendation alongside amitriptyline, to better reflect the balance between the benefits and harms associated with the 3 medicines. We also added links to relevant technology appraisal guidance in the section on treating and preventing migraine with or without aura. This is to provide easy access to relevant guidance at the right point in the guideline only and is not a change in practice.
Next review: This guideline will be reviewed if there is new evidence that is likely to change the recommendations.
How we prioritise updating our guidance
Decisions about updating our guidance are made by NICE’s prioritisation board. For more information on the principles and process, see NICE-wide topic prioritisation: the manual.
For information about individual topics, including any decisions affecting this guideline, see the summary table of prioritisation board decisions.
Recommendations
This guideline includes recommendations on:
See the 1-page visual summary on prophylaxis of migraine with or without aura.
Who is it for?
- Healthcare professionals who provide care for young people and adults with headaches
- Young people (12 years and older) and adults with headaches, and their families and carers. Particular consideration is given to the needs of people of reproductive age
Guideline development process
How we develop NICE guidelines
This guideline was previously called headaches: diagnosis and management of headaches in young people and adults.
Your responsibility
The recommendations in this guideline represent the view of NICE, arrived at after careful consideration of the evidence available. When exercising their judgement, professionals and practitioners are expected to take this guideline fully into account, alongside the individual needs, preferences and values of their patients or the people using their service. It is not mandatory to apply the recommendations, and the guideline does not override the responsibility to make decisions appropriate to the circumstances of the individual, in consultation with them and their families and carers or guardian.
All problems (adverse events) related to a medicine or medical device used for treatment or in a procedure should be reported to the Medicines and Healthcare products Regulatory Agency using the Yellow Card Scheme.
Local commissioners and providers of healthcare have a responsibility to enable the guideline to be applied when individual professionals and people using services wish to use it. They should do so in the context of local and national priorities for funding and developing services, and in light of their duties to have due regard to the need to eliminate unlawful discrimination, to advance equality of opportunity and to reduce health inequalities. Nothing in this guideline should be interpreted in a way that would be inconsistent with complying with those duties.
Commissioners and providers have a responsibility to promote an environmentally sustainable health and care system and should assess and reduce the environmental impact of implementing NICE recommendations wherever possible.