Overview
This guideline covers diagnosing and treating health-related fertility problems. It aims to reduce variation in practice and improve how fertility problems are investigated and managed.
For information on related topics, see our women's and reproductive health summary page.
Last reviewed: 31 March 2026
This guideline updates and replaces NICE guideline CG156 (February 2013).
Next review: This guideline will be reviewed if there is new evidence that is likely to change the recommendations.
How we prioritise updating our guidance
Decisions about updating our guidance are made by NICE’s prioritisation board. For more information on the principles and process, see NICE-wide topic prioritisation: the manual.
For information about individual topics, including any decisions affecting this guideline, see the summary table of prioritisation board decisions.
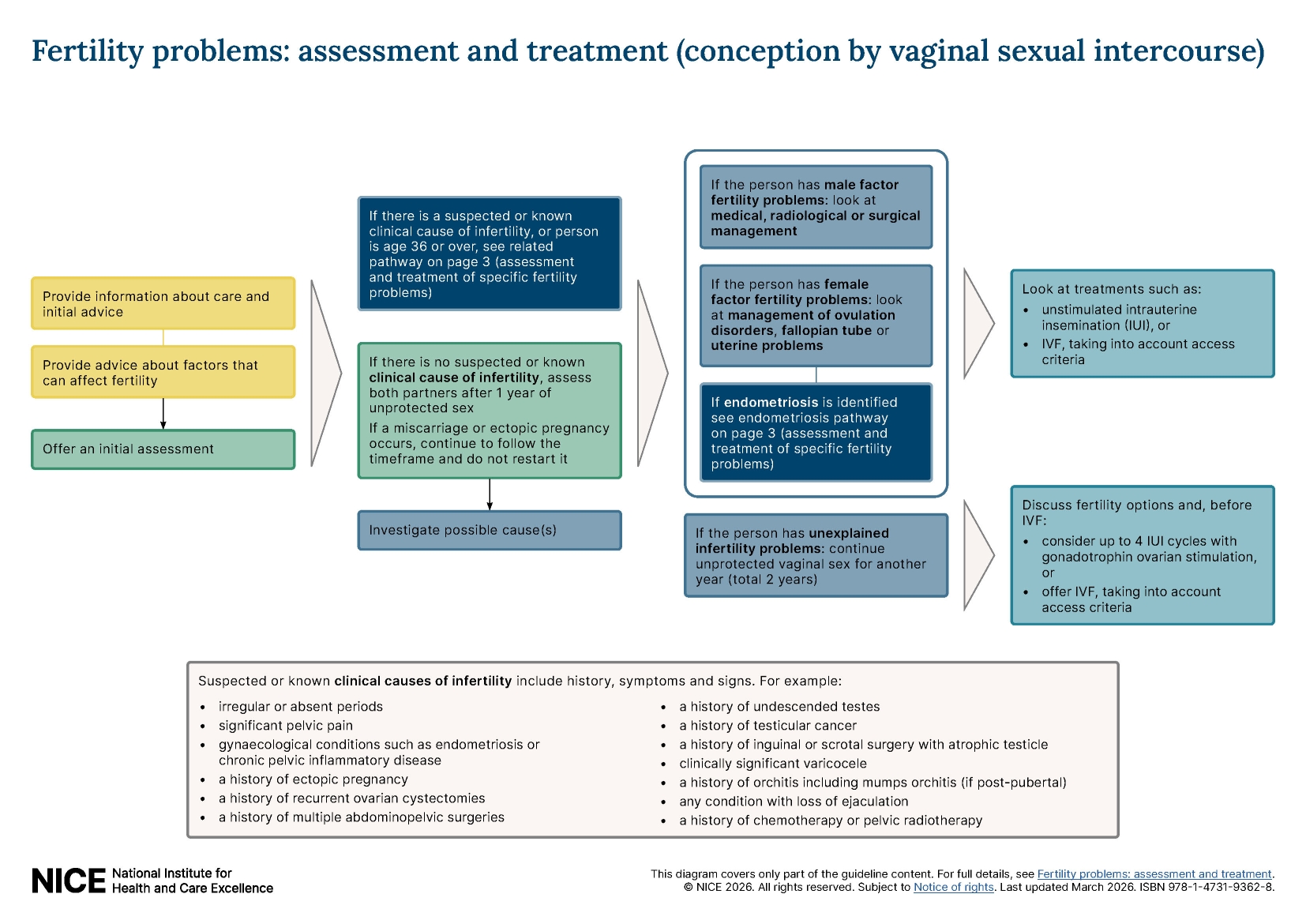
Recommendations
This guideline includes new and updated recommendations on:
- investigation of fertility problems and management strategies
- management of ovulation disorders, and tubal and uterine surgery
- unexplained fertility problems in people trying to conceive through unprotected vaginal sexual intercourse
- endometriosis
- access criteria for IVF treatment and procedures used during IVF
- intracytoplasmic sperm injection
- fertility preservation for medical indications
These supplement the existing recommendations on:
- principles of care and advice for people concerned about delays in conception
- investigating and managing fertility problems
- unstimulated intrauterine insemination
- donor insemination and oocyte donation
- long-term safety of assisted reproductive technologies.
Who is it for?
The recommendations apply to people who:
- have health-related fertility problems, or
- may need interventions to preserve fertility because they have a high risk of fertility problems because of clinical conditions, or medical or surgical interventions.
People with health-related fertility problems are those who:
- have a known clinical cause of infertility, or
- do not achieve a pregnancy:
- after 12 months of regular unprotected sexual intercourse, or
- after 6 cycles of artificial insemination.
The guideline applies to all people seeking assessment and treatment for health-related fertility problems who meet these criteria, regardless of their sexual orientation, partnership status or gender identity.
Guideline development process
How we develop NICE guidelines
Your responsibility
The recommendations in this guideline represent the view of NICE, arrived at after careful consideration of the evidence available. When exercising their judgement, professionals and practitioners are expected to take this guideline fully into account, alongside the individual needs, preferences and values of their patients or the people using their service. It is not mandatory to apply the recommendations, and the guideline does not override the responsibility to make decisions appropriate to the circumstances of the individual, in consultation with them and their families and carers or guardian.
All problems (adverse events) related to a medicine or medical device used for treatment or in a procedure should be reported to the Medicines and Healthcare products Regulatory Agency using the Yellow Card Scheme.
Local commissioners and providers of healthcare have a responsibility to enable the guideline to be applied when individual professionals and people using services wish to use it. They should do so in the context of local and national priorities for funding and developing services, and in light of their duties to have due regard to the need to eliminate unlawful discrimination, to advance equality of opportunity and to reduce health inequalities. Nothing in this guideline should be interpreted in a way that would be inconsistent with complying with those duties.
Commissioners and providers have a responsibility to promote an environmentally sustainable health and care system and should assess and reduce the environmental impact of implementing NICE recommendations wherever possible.